Russian soldiers showed their venal brutality when they caught a suspected Ukrainian spy on a motorcycle near a key air base.
Four masked military thugs held Ruslan Khammatov at gunpoint and marched him away for interrogation with a bag over his head. He is seen dressed in blue in a video of the troops taking him away.
They allegedly continued to torture him, using pliers in an attempt to rip off his ear.
The 40-year-old man was beaten, causing him to fracture his eye socket and ribs, as well as break his nose, while they tried to force a confession.
But they got the wrong man: they used their brutal torture against Putin’s loyal patron, while at the same time showing the bestiality of which they are capable.
Ruslan Khammatov, 40, was mistaken for a Ukrainian spy, but is actually a loyal supporter of Putin.
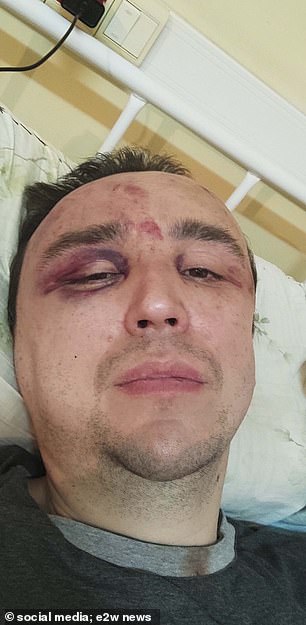

He was beaten, causing him to fracture his orbit and ribs, as well as a broken nose.
“They hit me on the forehead with a [gun] back, took out my ear with pliers and broke my ribs,” he revealed to the Russian media outlet Sota.
‘They threatened to rape me, put a stun gun in my anus and cut off my testicle.
“They found out he was divorced and said that meant he was gay.”
He said: ‘The first blow with a tactical glove hit me in the face, my nose was broken, I was bleeding, I covered my face and pressed myself to the ground, and then they kicked me.
“And they kept taking me out, talking to me, asking me questions, torturing me, and then this senior officer said, ‘You have two paths.’
‘The shortest way: now you tell everything as it is, you confess to us and we let you go, we don’t torture you.
‘And the second, they start to threaten you that we will cripple you, we will cut off your testicle.’
“They called it torture: ‘a stun gun in the anus, there will be blood, sweat, well, in short, total intimidation.'”

“They hit me on the forehead with a [gun] butt, he took out my ear with pliers and broke my ribs,’ he revealed to Sota

Khammatov is seen dressed in blue in a video of troops taking him away.
Kneeling in a pool of blood, he was forced to unlock his smartphone.
His protests of innocence were futile and he was falsely accused of making a hole in the fence of the Dyagilevo military airfield.
They found a Ukrainian channel on his phone, but it was staunchly pro-Russian.
The rest of the content on his cell phone was pro-war and showed that he supported dictator Vladimir Putin.
At that moment, realizing their mistake, they called the Russian traffic police, but not to report their own barbaric violence.
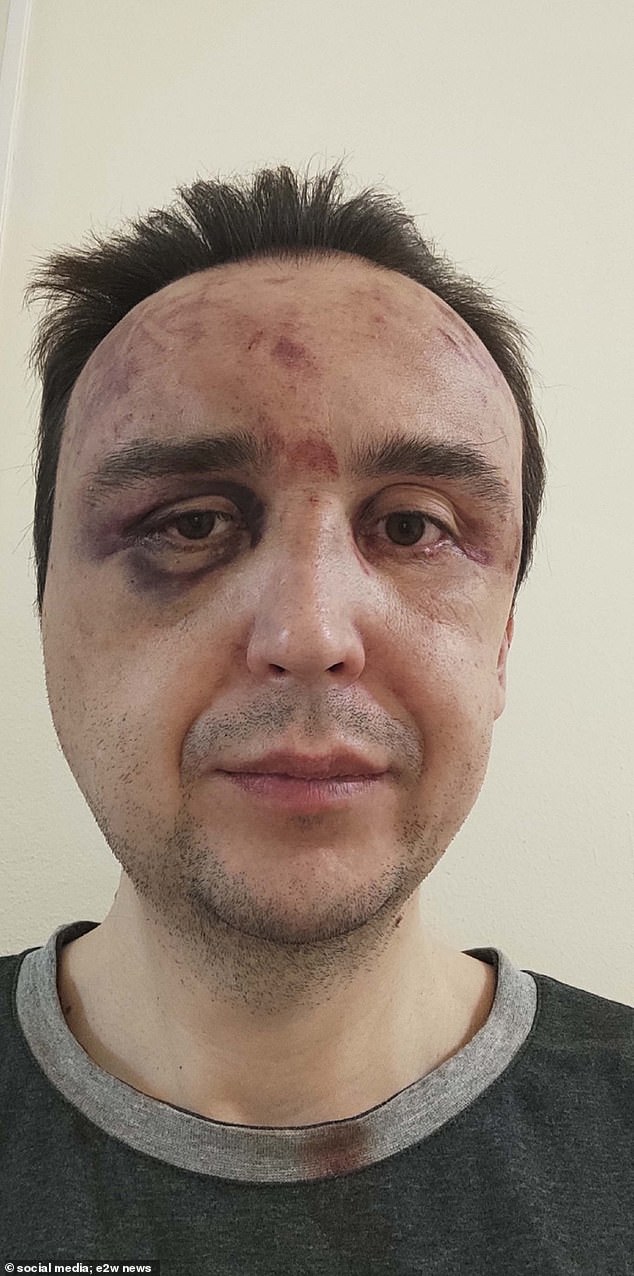
A blood stain is seen on Khammatov’s shirt after the ordeal. He reported that he was a victim of brutal torture.
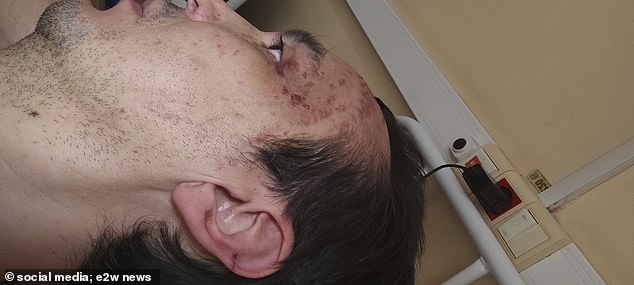
Wounds can be seen on Khammatov’s forehead and ear while he was receiving treatment.
They demanded that Khammatov lie to them that he had fallen from his motorcycle.
The police accepted the cover-up and fined the man for driving the motorcycle without a license plate, which he had just purchased and was testing when he was detained by the military torturers.
He was then loaded into an ambulance and sent to a hospital, which documented his injuries.

The content of his cell phone was pro-war and showed that he supported the dictator Vladimir Putin.
“I was detained by a group of four men in military uniforms and balaclavas,” said Khammatov, who is now seeking justice for the incident.
‘They presented themselves as a rapid response team, detained me and pointed Kalashnikovs at me.
‘They checked my documents and my phone number, took the motorcycle and escorted me.
‘They didn’t present me with any evidence of guilt, they beat me…’

