<!–
<!–
<!– <!–
<!–
<!–
<!–
The CDC has convened an urgent health panel on a possible bird flu outbreak in the United States, amid fears that the virus could soon spread to people.
Top agency officials met with local health leaders on Friday and told them to have “updated operational plans” in case H5N1 begins to spread among humans.
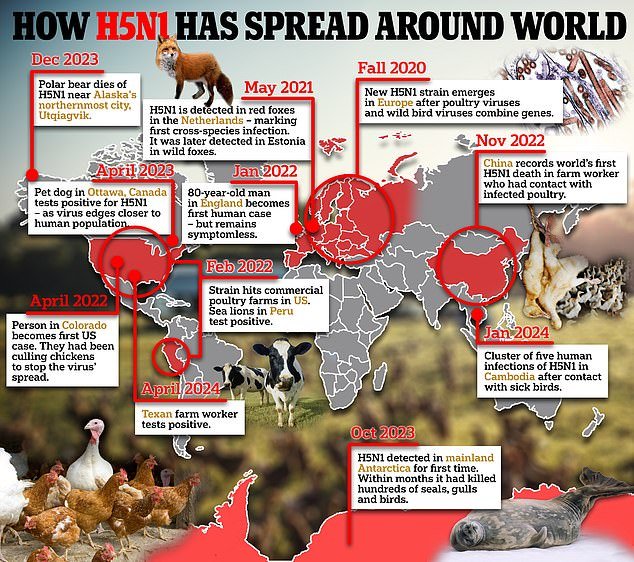
Only one case has been reported in one person in Texas so far, but doctors are now being urged to test other farmworkers if they suspect an infection.
Officials say the risk of bird flu to the public is low, but farm workers in close contact with infected animals and equipment are believed to be at higher risk.
The meeting was led by senior officials, including CDC Deputy Director Nirav Shah and its Director Dr. Demetre Daskalakis, who was involved in the national response to the monkeypox (or mpox) outbreak in 2022.
At the meeting, officials stressed that the risk to the public remained low but that local health leaders should be “prepared to respond.”
A CDC statement said: ‘CDC recommended that state public health officials…ensure they have updated operational plans to respond to avian influenza at the state level.
‘For example, CDC emphasized the importance of having plans to quickly test and treat potentially affected farmworkers following positive results among livestock herds.
“CDC also encouraged state health officials to communicate any challenges they face.”
The meeting was also attended by representatives of the Association of State and Territorial Health Officials, whose members include health officials from all 50 states, Washington, D.C., and five U.S. territories.
