Teenage girls may be a problem at the best of times, but a sinister new true crime series will delve into the more gruesome side of female rivalries, exposing the most heinous murders committed by young women.
Research discovery has given DailyMail.com an exclusive look at the trailer for her new Mean Girl Murders series, which offers insight into the sinister minds of slimy young assassins.
The second season of Mean Girl Murders will explore several twisted cases of anger and rivalry, lifting the lid on the darker side of female connections.
According to the network, the series will cover a series of shocking cases, including a love rivalry between a “dairy princess and a prom queen” whose fight over a handsome farmer “turns deadly.”

Investigation Discovery has released the trailer for its new Mean Girl Murders series: a look into the sinister minds of slimy young killers.
Mean Girl Murders Season 2 Will Explore Even More Twisted Cases of Rage
The documentary series will also shed light on a “fierce dispute between two top students for the title of valedictorian that results in a shocking act of violence in the middle of Economics class,” as well as a complicated case of how “a woman lies for there”. ‘Her own pregnancy motivates a twisted kidnapping case with a gruesome ending.’
“From ruthless competition to jealousy and cruel gossip, each episode will delve into what drove these bad girls to murder,” the synopsis reads.
Episode one of Deadly Harmony focuses on the gruesome 2012 murder of a Humble, Texas, mother who was found stabbed to death in the bed of her family home and the sexual relationship that was discovered between the victim’s daughter, Keri , and the murderer of his mother.
Mary Ann Murphy, 48, was attacked in her home around 1 a.m. on July 15, and her attacker broke in and stabbed her more than 70 times.
Murphy’s 16-year-old daughter, Keri, initially told neighbors and police that two men had broken in and attacked her mother before fleeing the scene.
However, by the time help arrived, his mother had died from her injuries.
‘(Mary Ann was stabbed) in the legs, in the arms, in the hands, in the neck, in the face,’ Craig Clopton, a former Harris County Sheriff’s Office investigator, said on a 2018 episode of Killer Couples. ‘It was obvious that it was a very brutal attack.’
“He said his mother was screaming loudly and I thought I heard a scream, but I wasn’t sure,” his neighbor Marcus Cox said at the time, the morning after the murder. “I thought they were street kids.”
It was just a few days before detectives realized that Kerri’s story was made up, and investigators became suspicious after noticing that the blood had clotted, indicating that Keri may have waited longer than she expected. He said to call the police.


The new season delves into more real-life mysteries that explore the dark side of female connections and rivalries.


Episode one of Deadly Harmony focuses on the gruesome 2012 murder of a Humble, Texas, mother who was found stabbed to death in her home.


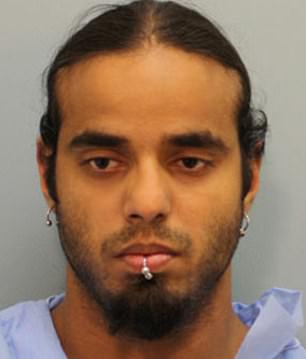

Charged: Rebecca Renee Keller (left), 20, and Zein Imran Ahmed (right), 21, were charged with the murder of Mary Ann Murphy in Texas; However, murder charges against Ahmed were dropped after it was discovered that Keller and Keri conspired and committed the murder.
Keri’s story began to change and after undergoing a polygraph test, the teenager confessed that she knew who killed her mother: her friend Zein Ahmed, 20.
Police arrested the couple after finding the knife used in the brutal stabbing in their home a few blocks away; However, they soon suspected a third person.
Their investigation into the phone calls and text messages between Kerri and Ahmed led them to Rebecca Keller, who was also Kerri’s older lover.
Kerri was a gregarious high school student and enthusiastic choir member until she met Keller, then 19, and was told by her friends and family that the teen then had a “crush” on the older girl.
‘They clicked immediately. It was an immediate attraction to each other,” Keri’s brother Scott recalled in an interview. Episode 2018 of Killer Couples.
“I’ve never seen Keri fall completely and utterly in love with a single person.” she added.


Other episodes this season of Mean Girl Murders will feature a rivalry between Dairy Princess and Homecoming Queen over a handsome farmer and a fierce dispute between two top students for the title of valedictorian that results in a shocking act of violence in the middle of class. Economy.
From ruthless competition to pangs of jealousy and cruel gossip, each episode will delve into what drove these bad girls to murder.
Keri confessed that she had Ahmed’s help in murdering her mother and that she didn’t approve of the age difference between her daughter and Keller, but it wasn’t long until they realized something wasn’t right.
Investigators examined phone records from a burner phone found in the family home and discovered that she had managed to continue talking to Keller after her mother forbade her from using the phone, and that they had been in constant communication on the night of the murder.
‘Keri was Googling ‘what’s the quickest way to kill someone?’ Clopton said. ‘Those Google searches were done at the same time she was on the phone with Rebecca. So I know you’re plotting this together.’
Keller, 20, was arrested and charged with murder, as well as sexual assault of a child, as she was in a sexual relationship with 16-year-old Kerri at the time.
Both girls were arrested and had phone calls recorded before going to trial and discovering that Keller stabbed Murphy, not her daughter. However, she was the mastermind behind the plan.
Both Keller and Keri accepted plea deals, with Keller sentenced to 60 years and Keri to 30. Murder charges again against Ahmed were dropped.
New episodes of Mean Girl Murders will premiere Monday, March 25 at 9/8c on ID.
