Taiwan has been hit by its strongest earthquake in 25 years, killing at least nine people, toppling buildings and even triggering tsunami warnings.
But amid the chaos, eerie images show the island’s tallest building, the $1.8 billion Taipei 101 skyscraper, swaying gently like a stick in the breeze.
The building, which measures 1,671 feet tall, is equipped with an ingenious solution that reduces the building’s overall sway by 40 percent during earthquakes and winds.
A 660-metric-ton golden sphere called a “tuned mass damper” hangs inside the building from the 92nd floor.
As the building moves in one direction, the steel sphere swings in the other direction and maintains the overall balance of the building.

The key to Taipei 101’s impressive structural integrity is a 660-metric-ton golden sphere hanging from the 92nd floor.
If wind or earthquake force pushes the tower to the right, the sphere will provide an immediate and equal force to the left, canceling the initial motion.
So even though the tower sways, it does not fall.
It is what is known as a “passive damping system”, which means that it works without any external energy or control, just gravity and the movement of the building.
Meanwhile, hydraulic pistons beneath the huge sphere absorb and dissipate energy as heat.
Dr Agathoklis Giaralis, professor of structural dynamics at City University of London, described the clever spherical device as “like a pendulum”.
“This steel sphere rests on damping devices that are designed to dampen the relative rolling motion between the structure and the sphere, acting in a similar way to shock absorbers in car suspensions,” he told MailOnline.
Although it is strange to see a building swaying, modern skyscrapers are built to be flexible, especially in earthquake-prone areas like Taiwan.
“The materials they are made from are elastic, meaning they stretch or contract depending on the changing loads acting on them,” Professor Antony Darby from the Department of Architecture and Civil Engineering at the University of Bath told MailOnline.
‘The amount of this deformation in an individual element (for example, a beam or a column) is very small, but when multiplied across all the elements of a tall building, this can cause significant lateral movements.
“These movements are not dangerous to the structure itself, but if excessive, they can cause discomfort to the occupants.”


Taipei 101 is the tallest building in Taiwan and formerly the tallest building in the world (now the 11th tallest)


The key to the building’s astonishing structural integrity is a 660-metric-ton golden sphere hanging from the 92nd floor: the “tuned mass damper.”


In Taiwan, Taipei 101 must withstand earthquakes and typhoons. Every eight stories, steel “stabilizing trusses” running from the core of the building to the exterior columns were installed in the building to increase the rigidity of the towers, and to prevent them from swaying and vibrating during an earthquake, a huge tuned mass damper: A device for transferring energy from tower motion to giant dampers. This 660-ton ball of stacked steel plates hangs from steel cables from the 92nd floor of the tower. It is connected to the building structure via eight liquid-filled dampers.
Construction of Taipei 101 began in 1999 and was completed in time for New Year’s Eve 2004, when it finally opened to the public.
It was designed to look like a bamboo shoot that rises upward in eight sections, looking like a series of square cubes stacked on top of each other.
Each floor was installed with steel “outrigger beams” running from the core of the building to the exterior columns to increase the rigidity of the towers.
While other tall buildings keep tuned mass dampers hidden, Taipei 101 is visible to visitors from the 88th to 92nd floors, making it something of a tourist attraction.
Unfazed tourists have even filmed the sphere’s movement during previous earthquakes, which are frequent on the island.
Taiwan is a country especially prone to earthquakes because it is near where two tectonic plates meet: the Philippine Sea plate and the Eurasian plate.
Powerful seismic activity can be detected along the edges of tectonic plates, where plates rub against each other and cause earthquakes.
However, Taiwan’s earthquake preparedness is “among the most advanced in the world,” according to Stephen Gao, a seismologist and professor at Missouri University of Science and Technology.
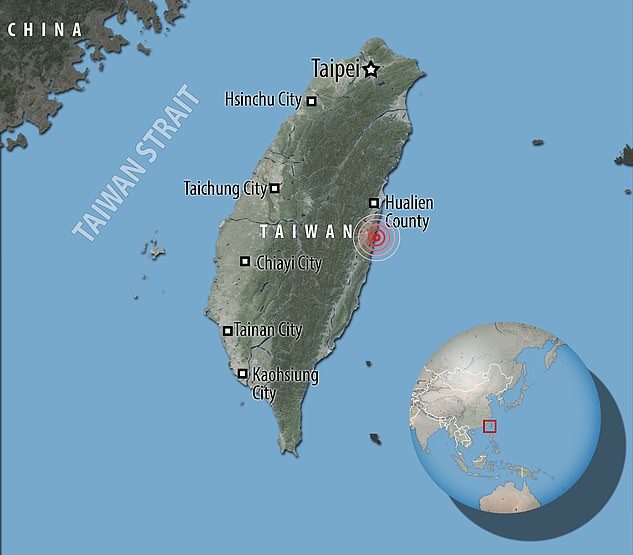

The powerful earthquake struck off the east coast of Taiwan on Wednesday morning. Taiwan is a country especially prone to earthquakes because it is close to where two tectonic plates meet.


This photo taken by Taiwan’s Central News Agency (CNA) on April 3, 2024 shows a damaged building in Hualien, after a large earthquake hit eastern Taiwan.
“The island has implemented strict building codes, a world-class seismological network and extensive public education campaigns on earthquake safety,” he said.
At the time of writing, the strongest earthquake to hit Taiwan in 25 years has killed nine people, while another 50 are missing.
Rescuers expect the total number of injured and dead to rise as the search for people trapped by the earthquake continues.
The earthquake and its aftershocks also caused 24 landslides and damaged 35 roads, bridges and tunnels, according to government statistics.
Taiwan’s earthquake monitoring agency said the quake had a magnitude of 7.2, while the United States Geological Survey (USGS) estimated it at 7.4.