Swarms of AI-enabled killer drones may seem like the plot of a dystopian sci-fi thriller.
But in a terrifying vision of the future, a scientist has shown how easy it already is to build a “killer drone” that can hunt and kill people.
In just a few hours, Luis Wenus, an engineer and entrepreneur, turned a $115 (£89.99) drone into the basis of a deadly weapon.
Using AI facial recognition, the drone was programmed to recognize individuals and race towards them at full speed.
Although Wenus says he built the drone “for a game,” he also says he wanted to raise awareness about how easily it could be used in a deadly terrorist attack.

Killer drones were the inspiration for the 2019 sci-fi horror film The Drone, but they may be closer to reality than we thought and even easier to make than we thought.
In a video posted on X, formerly Twitter, Wenus and his fellow engineer demonstrated how a commercial drone can be programmed to pursue targets.
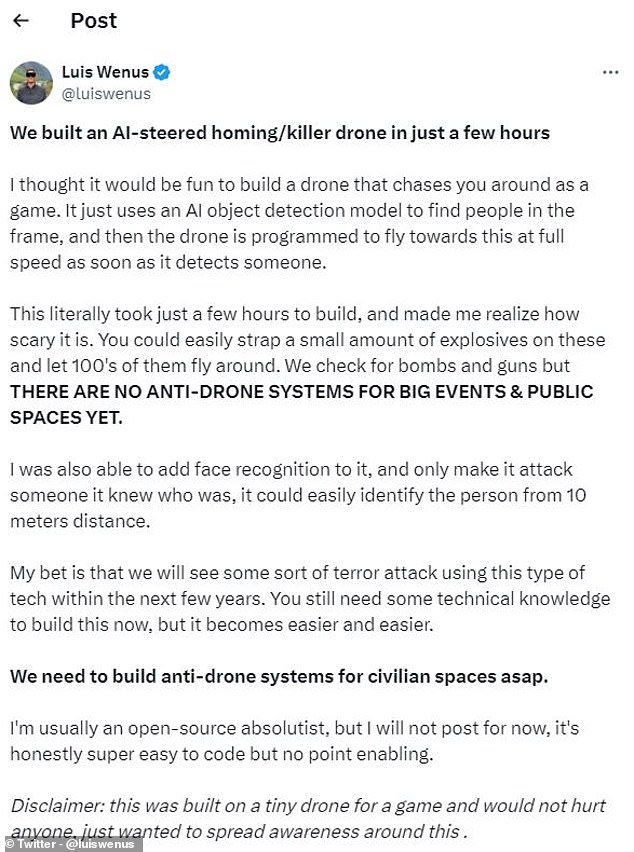
In the post, Wenus writes: “I thought it would be fun to build a drone that would chase you around like a game.”
However, the terrifying video quickly shows how dangerous this technology could really be.
The drone uses an AI ‘object detection model’ to recognize faces using its built-in camera.
Once it sees a face, the drone is programmed to try to keep it in the center of its vision and fly directly forward, chasing its target.
In one mode, the drone will charge directly toward anything it recognizes as a face.
In this mode, the video shows how the drone begins to “attack” other people in the park and only stops when Wenus catches it.


Luis Wenus, engineer and entrepreneur, showed how he turned a commercially available drone into a ‘killer drone’ designed to hunt targets in just a few hours
The drone also has a second configuration that Wenus describes as a “murder drone.”
Wenus wrote: “I was also able to add facial recognition to it and only have it attack someone who knew who it was; I could easily identify the person from 10 meters away.”
The video shows how the drone selectively chooses to pursue its target, ignoring other people.
And while this drone isn’t particularly dangerous, Wenus warns that more deadly drones could pose a serious threat.
He wrote: “This literally took only a few hours to build and made me realize how scary it is.” You could easily put a small amount of explosives on them and let hundreds of them fly around.’

In a post on X, formerly Twitter, Wenus says he wanted to develop the drone “as a game,” but quickly realized how dangerous the technology could be.


The video shows how the drone is programmed to recognize faces and fly at full speed towards them

On social media, some commentators expressed a mix of admiration for the technology and fear for what it might be capable of.

Other commentators were concerned that developing killer drones would now be extremely easy with a little technical knowledge.
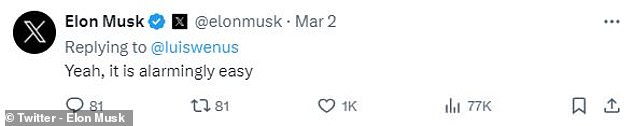
Even Elon Musk, CEO of X, expressed concern that it is “alarmingly easy” to create autonomous killer drones.
On social media, commentators reacted with a mix of fascination and horror at the invention.
In response to the post, one X user wrote: ‘On a surface level this is cool, but the potential to use it for evil is a little scary!’
Another commenter added: “Yeah, that shouldn’t be easy to build…”
Even Elon Musk, CEO of X, responded to the post by saying, “Yes, it’s alarmingly easy.”
Small explosives-carrying drones are already having a significant impact on the war in Ukraine.
Ukrainian drone operators attach grenades, mortars and other explosive devices to cheap, commercially available drones to create deadly weapons.
These first-person drones have proven extremely effective at hunting down and destroying everything from individual troops to tanks and bunkers.
But the crucial difference is that these drones are operated by an individual who is always in control of the process rather than an autonomous AI.


Ukrainian drone operators (pictured) have been using cheap commercial drones with explosive devices to hunt Russian tanks and bunkers.
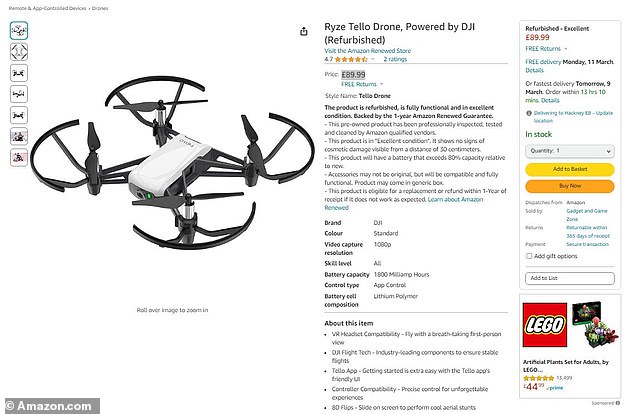

The drone used by Wenus in this demonstration is extremely cheap and easy to obtain, as it is available on Amazon for just $115 (£89.99).
The real danger is that swarms of cheap and easy-to-produce autonomous drones could be launched onto battlefields or even public spaces.
Wenus writes: “My bet is that we will see some type of terrorist attack using this type of technology in the next few years.
“It still takes some technical know-how to build this now, but it will get easier.”
Autonomous drone swarms have not yet been deployed on the battlefield, but DARPA is reportedly developing a drone swarm weapon of mass destruction called AMASS (Autonomous Multi-Domain Adaptive Swarm Swarms).
AMASS is still in the planning stages, but DARPA (Defense Advanced Research Projects Agency) has been collecting bids from vendors for the $78 million contract.
Wenus wrote: ‘We check for bombs and guns, but THERE ARE STILL NO ANTI-DRONE SYSTEMS FOR LARGE EVENTS AND PUBLIC SPACES. “We need to build anti-drone systems for civilian spaces as soon as possible.”
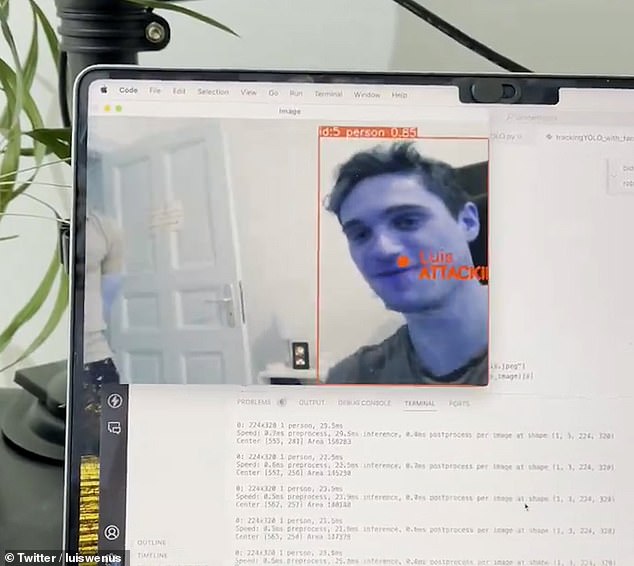

Wenus demonstrates the drone’s “kill mode.” This image shows how the drone recognizes his face and identifies him as a target with the instruction ‘ATTACK!!’
Wenus describes himself as an “open source absolutist,” meaning he believes that major advances in code, and in AI in particular, should be open to the public.
And, in responses to the post, several social media users asked Wenus to post the code or share a full tutorial on how to produce a killer drone.
However, in this case it says it will refrain from publishing the code due to the potential danger posed by autonomous drones.
“I won’t post for now. Honestly, it’s very easy to code, but there’s no point in enabling it,” he added.
