The UK is entering a “cancer renaissance” with pioneering personalized vaccines to combat some of the deadliest tumors expected by 2030, leading experts say.
Trials are already underway offering patients with lung, liver, kidney, bowel and pancreatic cancer injections that could prevent the disease from coming back.
A first-of-its-kind partnership between the NHS and cancer vaccine developers, announced last year, will mean British patients will have priority access to these breakthrough drugs.
The personalized injections, created using the same technology behind Covid vaccines, train the body to hunt cancer cells and prevent the disease from returning.
However, there are question marks over the affordability of the treatment, which costs around £400,000 per patient, inside sources told MailOnline.
Steve Young, 52, from Stevenage is one of the first patients in the melanoma vaccine trial at University College Hospital, London.
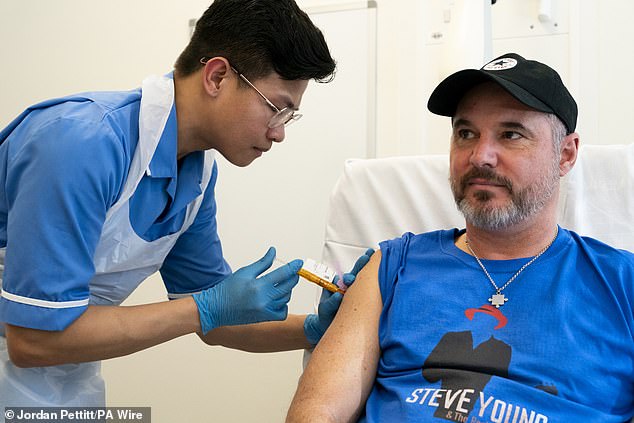
Steve receives his first melanoma injection at University College London Hospital from nurse Christian Medina.
The news comes after today’s announcement that the first personalized melanoma skin cancer vaccine was being trialed in NHS patients.
Early results suggest the vaccine can dramatically improve the chances of survival for patients with this disease, the deadliest form of skin cancer.
One patient who has already received the vaccine, Steve Young, 52 from Stevenage, described it as his “best chance of stopping cancer in its tracks”.
Mr Young was diagnosed with melanoma after a bump on his head, which he believes he had had for about a decade, turned out to be cancerous.
However, experts say that melanoma is just one of many cancers that could be cured thanks to personalized vaccines.
“These vaccines could help cancer patients across the board,” says Dr. Lennard Lee, a cancer vaccine expert at the University of Oxford.
‘This is a new technology that we didn’t have before and that could save patients’ lives.
‘There are already trials underway for head and neck, bowel, pancreas, lung and liver cancers, and the number will only increase month by month, year by year.
‘It’s a whole field of medicine that has emerged from nowhere. “It looks like the UK is entering a cancer treatment renaissance.”
Professor Lee added: “All drug manufacturers are targeting 2030 as the year to commercialize these vaccines.”
Cancer vaccines are designed to target specific genetic mutations found in cancer cells.
This means that if the cancer comes back, the immune system will detect it immediately and destroy the cells before they have time to spread.
To create the injection, a sample of the tumor is removed during the patient’s surgery.
It is then sent to a laboratory where the tumor’s genes are sequenced to identify certain proteins created by the cancer cells, known as neoantigens, that will trigger an immune response.
They are then used to create an individualized mRNA vaccine, the same cutting-edge technology used to develop several of the Covid shots.
The vaccine tells the patient’s body to generate tumor-specific neoantigens that in turn train the immune system to recognize and attack tumor cells.
Data published last year found that, in a small study, high-risk melanoma patients who received the new shot along with another immune-boosting drug were almost half as likely to die or have their cancer return after three years, compared to those only taking immunotherapy.
Patients must receive a new injection, along with a course of immunotherapy, every three weeks for a year.
However, experts say they are particularly excited about the vaccines’ potential to help cure patients with pancreatic cancer, a deadly form of the disease with few effective treatments.
Each year, around 10,000 patients are diagnosed with pancreatic cancer and approximately the same number die annually from it.
The condition is so deadly because it is often diagnosed late due to a lack of obvious symptoms.
Only about five percent of patients will survive the disease for more than a decade.
Earlier this month, new data from a study that gave mRNA vaccines to pancreas patients after surgery showed signs that it induced a strong immune response that would reduce the risk of the disease returning.
A larger study is currently being carried out, involving 260 patients.
“We have made great progress in treating melanoma over the years and can cure around half of all patients,” said Professor Tom Powles, director of Barts Cancer Centre.
‘But there is nothing that works for pancreatic cancer.
‘If given to patients early enough, cancer vaccines could open a new chapter for pancreas patients by reducing the risk of the disease returning.
“If I were a pancreatic cancer patient, this is the trial I would want to participate in.”
However, there are concerns about the cost of the vaccine.
‘It currently costs around $500,000 [£399,000] to develop each cycle of injections,” says an industry source.
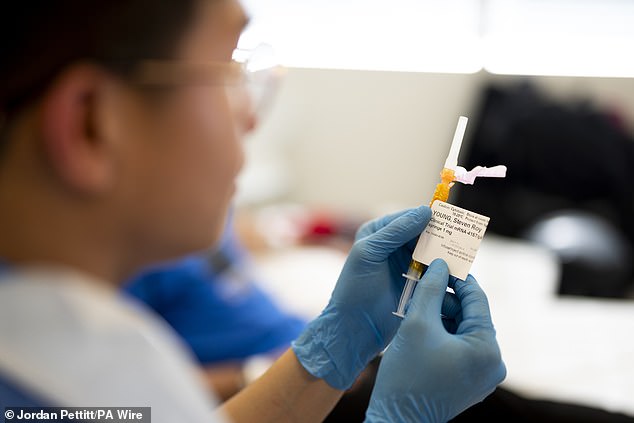
The shots use the same mRNA-based technology found in the Covid vaccine to harness the body’s immune system and kill cancer.
‘At that price, the NHS will struggle to afford it.
“Pharmaceutical companies will have to find a way to make vaccines commercially viable.”
But experts say the NHS’s partnership with vaccine developers Moderna and BioNTech, which will see thousands of UK patients enrolled in their trials, could allow Britons to access these new treatments sooner than those in other countries.
“In the coming years, NHS cancer patients can expect to see more opportunities to participate in these vaccine trials,” says Dr Lee.
“These are opportunities that patients elsewhere might not have.”


