Britons will be able to buy Covid jabs privately from Boots from next week.
The jabs were previously only available on the NHS as part of the national response to the pandemic and preparations for winter.
But in a major shake-up of the plan, the high street chain will begin selling single-dose Pfizer vaccines in 52 stores in England from April 1.
But if you want one, you’ll have to pay the eye-watering cost of £98.95.
Brits aged 12 and under will be able to start booking appointments later this week, MailOnline understands.

The retail chain will sell single-dose Pfizer vaccines in 50 stores in England, at an incredible cost of £98.95. Brits aged 12 and under will be able to start booking appointments later this week, MailOnline understands.


Individual stores will release appointments this week to begin in early April. MailOnline understands the price will allow Boots to cover the cost of the vaccine and other operational costs involved in providing the service.

Pfizer was one of the big winners of the pandemic. As lives and businesses crumbled amid lockdowns and disruptions to life during Covid, the New York-based company became a household name.
Experts have long called for Covid vaccines to be sold privately, since the immediate threat of the virus began to fade.
Pfizer was one of the big winners of the pandemic. As lives and businesses crumbled amid Covid shutdowns and disruption, the New York-based company became a household name.
During the height of the pandemic, adults and certain groups of children in the United Kingdom were eligible to receive vaccines against the virus.
Officials were desperate to get as many people as possible to take the jabs to build a wall of immunity to keep Covid at bay and allow the country to end the series of crippling lockdowns.
But the rollout has changed course in recent fall and spring booster rollouts.
The jabs once available to almost everyone have been restricted to at-risk Britons such as care home residents, the over-65s and frontline NHS workers who normally come into contact with them.
As such, millions of previously vaccinated Britons have not received a booster dose since late 2021.
Experts say offering Covid vaccines privately, as happens with the flu every winter, is a no-brainer.
Even a member of the Government’s own vaccine advisory panel said it was a “good idea” ahead of its rollout in autumn 2023.
A Boots spokesperson said: ‘We are launching a private Covid vaccination service for people who are not eligible for an NHS vaccine but still want the option to protect themselves from the virus.
“Our private service builds on our current delivery of Covid vaccines for the NHS and we are pleased to now be able to offer Covid vaccines both on behalf of the NHS and privately, as we have done with flu vaccines for many years.”
Individual stores will release appointments this week to begin in early April.
MailOnline understands that the price of almost £100 allows Boots to cover the cost of the vaccine and other operational costs involved in providing the service.
The Pfizer vaccine is attractive to non-NHS providers because it can come in a single-dose vial, meaning pharmacies do not have to prepare multiple shots at once.
It uses the same formulation that UK regulators approved in September.
In February, Pharmadoctor, a clinical services provider working with more than 8,000 pharmacies across the UK, announced it would be offering private Covid vaccines from April 1 for just £45.
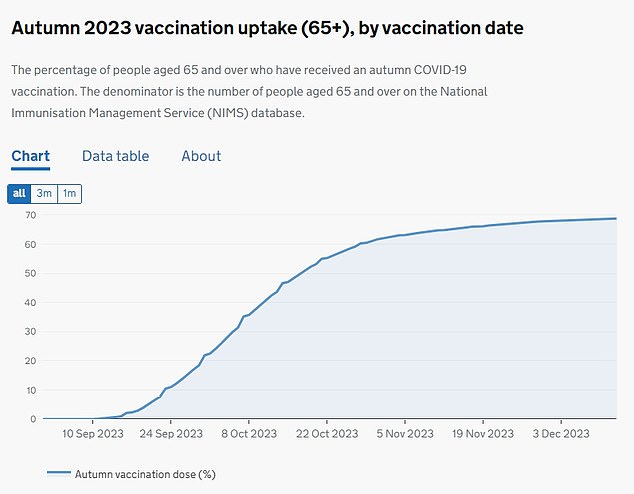

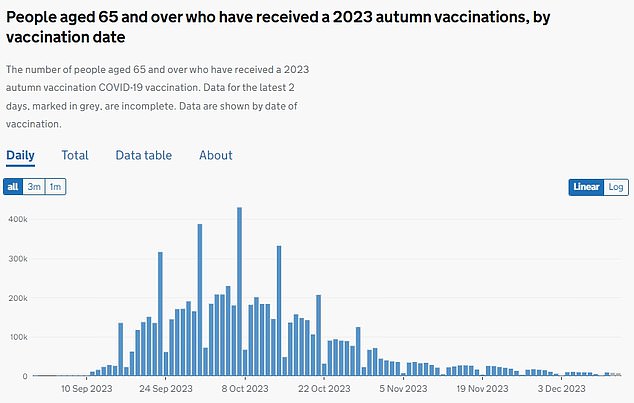

Acceptance of the autumn booster rollout stood at around 68.8 percent (pictured). As of December 2023, 7.8 million people had received a shot under the program.
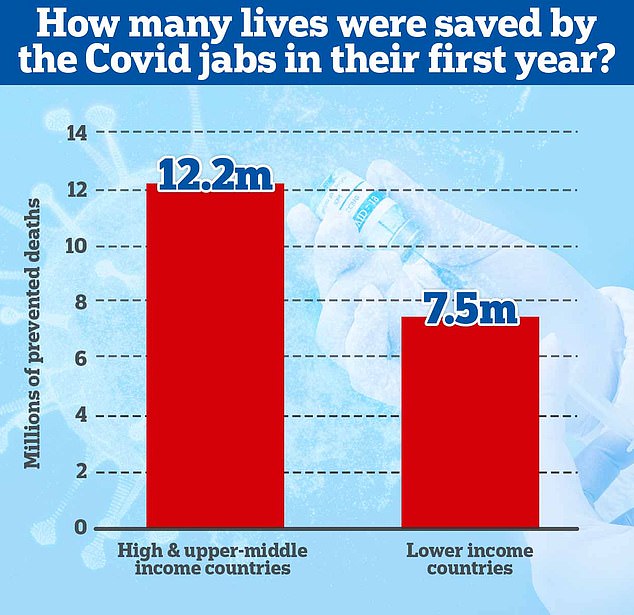

A 2022 study led by academics at Imperial College London suggests that Covid vaccines saved almost 20 million lives in the first year since countries began rolling out the shots, mostly in wealthy nations.
The supplies from its private offering will be the Novavax and Pfizer vaccines.
Moderna, another pharmaceutical giant that made a Covid mRNA like Pfizer’s, has already said it is looking to offer its vaccine privately.
Ministers paid around £20 per Covid jab to Pfizer during the height of the pandemic for its jabs.
But in August Moderna He said he expects to quadruple his own price when the jabs are offered privately.
It comes as the Joint Committee on Vaccination and Immunization (JCVI), which makes recommendations on the vaccine rollout to the Government, set out last month that the spring booster campaign due to launch in mid-April would be further restricted.
The latest rollout will only offer shots to adults 75 and older, seniors in nursing homes, and immunocompromised children and adults.
Health officials have also indicated that eligibility for the NHS campaign in the autumn will also likely be reduced to save money.
