<!–
<!–
<!– <!–
<!–
<!–
<!–
Top US health officials are making plans to roll out a new, updated Covid vaccine this winter.
The Food and Drug Administration (FDA) revealed Monday that it will hold a public meeting in May to discuss whether the current crop of vaccines needs to be updated and which variant should be included in next winter’s dose.
Currently, the most common strain is JN.1, which was not targeted by the previous booster that inoculated against XBB.1.5, although there is still some cross-protection.
The agency said: “The FDA anticipates that changes to the vaccine composition may be necessary based on the vaccine strains currently circulating.” [Covid] virus.’
Officials believe the Covid vaccine will need to be updated every year, as with flu vaccines, to bolster protection against emerging variants.
The United States is deciding the composition of next winter’s Covid vaccine (file photo)
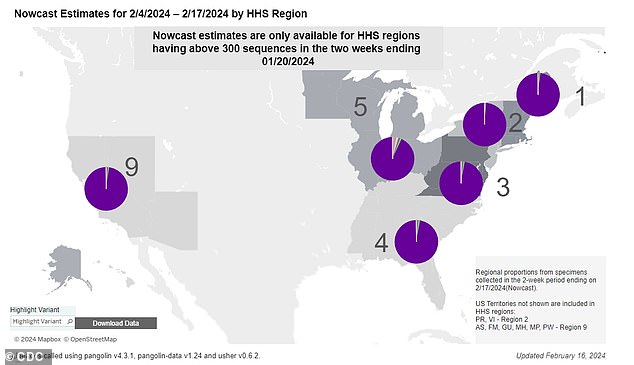

The currently dominant variant is JN.1, which is behind almost all cases (shown as the purple area in the pie charts for each US region).
The vaccine is likely to be recommended for all Americans over six months old, even though many experts maintain it is only necessary for people 75 and older or those with underlying conditions.
It comes amid growing skepticism about vaccines across the United States.
Data shows that only forty percent of people 75 and older, who are most at risk of contracting the virus, received the updated vaccines last year. Among those over 18 years of age, only 20 percent showed up to receive the new Covid vaccine.
The FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC), a 16-member committee that decides the composition of vaccines, will hold the meeting.
The agency added: “Barring major new changes in the circulating virus, the FDA expects that the composition of Covid vaccines will need to be updated annually, as is done with the seasonal influenza vaccine.”
The committee is not expected to make a recommendation until June of this year, and then vaccine companies like Pfizer and Moderna will be asked to update their vaccines.
They do this by changing the Covid spike protein, or virus protein used to invade cells, that their mRNA vaccines cause cells to develop, training the immune system to recognize a new variant.
Like last year, the vaccines are not expected to receive federal funding and will instead be covered by the private market.
It is unclear what level of effectiveness against hospitalization new vaccines may need to show in trials, but for the flu vaccine, which is updated each year, this may be as low as 40 percent in some years.
The FDA and the Centers for Disease Control and Prevention (CDC) will then approve the new vaccines, which will likely be rolled out in September.
Officials hope to update Covid vaccines annually to match a new variant, similar to how flu vaccines are updated.
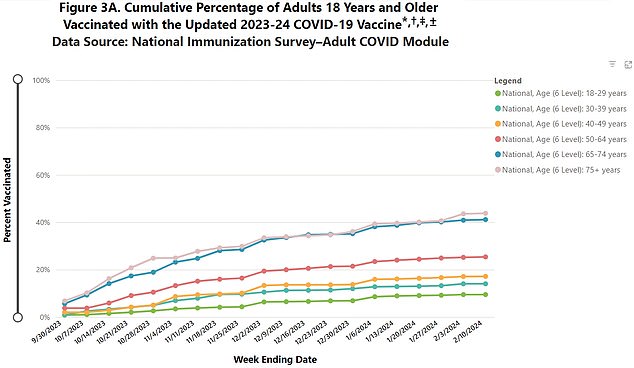

The graph above shows uptake of last year’s Covid booster dose by age group, with those over 75, who are also at highest risk, most likely to receive the vaccine.
The same committee also updates the flu vaccine and makes its recommendation based on the flu variant dominant in the Southern Hemisphere, which has its winter when the United States has its summer.
The vaccines reduce the risk of hospitalization or death from Covid by more than 90 percent, although they may not be able to prevent someone from developing an infection.
Immunity works against new Covid variants because of “cross-protection” – the immune system can still recognize other proteins in the new variant and launch a response.
Covid cases in the US are currently declining and the Covid test positivity rate (proportion of tests that detect the virus) is now 8.1 percent nationwide over the past week, in compared to 9.4 percent the previous seven days.
Hospitalizations are also trending downward, with 18,977 new admissions recorded last week, down 12 percent from 20,100 seven days earlier. And deaths are also falling.
