- Ticket prices from Sydney to Melbourne have soared
- Qantas economy tickets cost over $1,000
- Australian Grand Prix once again expects record attendance
<!–
<!–
<!–
<!–
<!–
<!–
The cost of flights to Melbourne for the upcoming Australian Grand Prix has soared, with some fans now saying they would prefer to take the train or drive to the event to avoid incredible expenses.
The third round of the Formula 1 championship will take place this Sunday in Melbourne, with two local drivers on the grid for the first time in more than a decade.
A record 444,631 F1 fans attended last year’s event at Albert Park, making it Melbourne’s busiest weekend sporting event.
Ticket sales for this year’s race are booming and previous records are expected to be broken in a new milestone for the race.
However, some fans online have complained about the incredible price of plane tickets from Sydney to Melbourne.

Flights to Melbourne for the Australian Grand Prix have soared (pictured, world champion Max Verstappen on his way to winning last year’s race at Albert Park)


Some fans said they would rather drive or take a train to the event this weekend due to the incredible price of plane tickets (stock image)
An X user, who also claims to be a pilot, posted about the situation on Wednesday.
“The F1 GP once again pushes the capacity of the MEL-SYD air route to its limits,” he wrote.
“Good luck getting an economy seat for less than $1,000.”
“The market is strained these days as the 330 QF fleet does not have the capacity to cover a few MEL-SYD routes. »
The user then explained that Virgin Australia and Rex were already sold out.
For context, one airfare comparison website listed the average ticket price from Sydney to Melbourne as $166.
Daily Mail Australia checked the price of Qantas return economy tickets from Sydney to Melbourne for the grand prize and found they cost up to $1,030.
‘Take the XPT, cheaper!! It doesn’t matter how long it takes to travel, what matters is the money saved at this point,” one user replied.
“So drive?” it will probably cost $300-$400 in fuel (round trip),” another wrote.
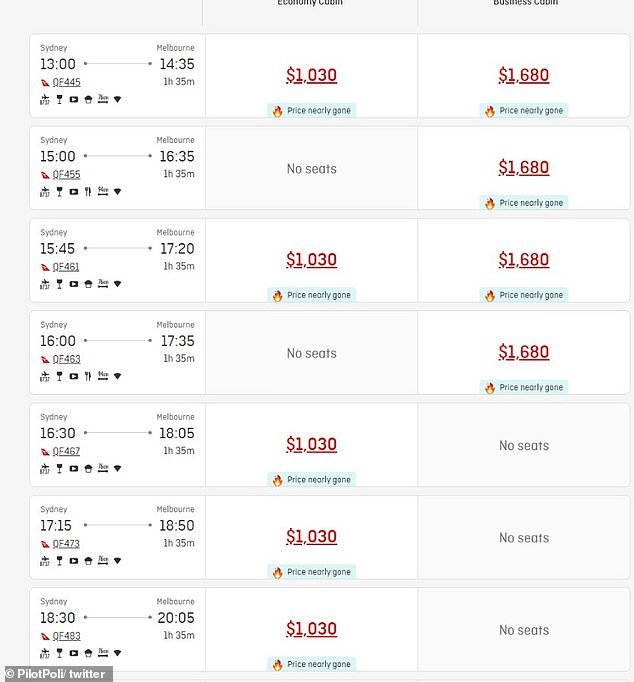

Qantas economy tickets to Melbourne currently cost over $1,000
“Imagine how efficient a fast train would be,” said a third.
Australian Grand Prix chief executive Travis Auld said the presence of two Australians on the grid – Oscar Piastri and Daniel Ricciardo – was one of the main reasons for the rush for tickets this year.
“It certainly makes life easier when you have two drivers on the grid and it increases fan interest,” he said.
“There are fans who have their teams and their drivers, and certainly the more casual fans embrace the Australian drivers very quickly and they don’t just make up numbers.”
