- Trump visited José Alba, a winemaker who killed an ex-convict in self-defense
- The former president made the stop after the second day of his criminal trial
- Trump received a raucous reception when he arrived at the winery on Tuesday.
Donald Trump left his criminal trial Tuesday and visited a warehouse worker acquitted of murder for stabbing an ex-convict to death in self-defense at his Manhattan store.
The former president headed to Hamilton Heights to speak with José Alba, who stabbed Austin Simon to death in July 2022, when he jumped over the counter of his store.
Trump received a raucous welcome when he arrived at the winery, and the crowd chanted “four more years” as the former president joined in.
The charges against Alba were dropped in July by Manhattan District Attorney Alvin Bragg, who has become a frequent target of Trump’s attacks during the election campaign.
Alba killed Austin Simon with a knife in July 2022, when he jumped over the counter of his store.
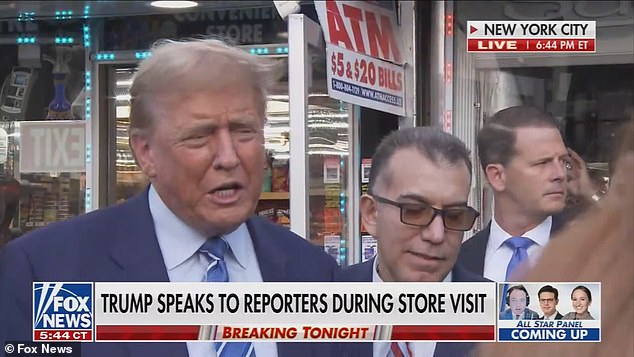
Trump received a raucous reception when he arrived at the warehouse on Tuesday, as a crowd chanted “four more years” for the former president.

Alba (right) fled to upstate New York and planned to return to his home in the Dominican Republic because he feared for his life after being charged.
Alba was attacked by Simón’s girlfriend during a fierce dispute over a bag of chips, when she tried to pay $3 but her card was declined.
While Alba and Simon were fighting, the girlfriend who started the fight pulled a knife from her purse and slashed Alba on the arm.
The girlfriend was never charged, but Alba was charged with murder in a case that sparked fury throughout the city.
Alba fled to upstate New York and planned to return home to the Dominican Republic because he feared for his life after being charged, and he quit his job after receiving an outpouring of hate over the stabbing.
But now he’s back in town.
On July 19, Manhattan District Attorney Alvin Bragg’s office asked a judge to drop charges against Alba after she faced intense backlash, including criticism from the city’s mayor.
Trump’s appearance came at a time when Bragg has been a frequent target of his attacks.

Trump stopped at the warehouse at the conclusion of the second day of his criminal trial

The girlfriend was never charged, but Alba (above, entering a police station) was charged with murder in a case that sparked a citywide furore.
Surveillance footage clearly showed that Simon had moved behind the counter at the convenience store in Hamilton Heights and pushed the store clerk into a stack of shelves before grabbing him.
It was only then that Alba approached a shelf and pulled out a knife to defend herself.
During the altercation, Alba was apparently stabbed by Simón’s girlfriend, who instigated the argument when her benefits card was declined. The girlfriend was never charged for the incident.
The motion to dismiss said prosecutors “have determined that we cannot prove beyond a reasonable doubt that the defendant was not justified in his use of deadly physical force.”
The document said that “a murder case against Alba could not be proven at trial beyond a reasonable doubt” and that the case will not be presented to a grand jury.

Video shows that warehouse worker José Alba was trying to avoid confrontation with Austin Simon in the minutes before the July 1 stabbing.
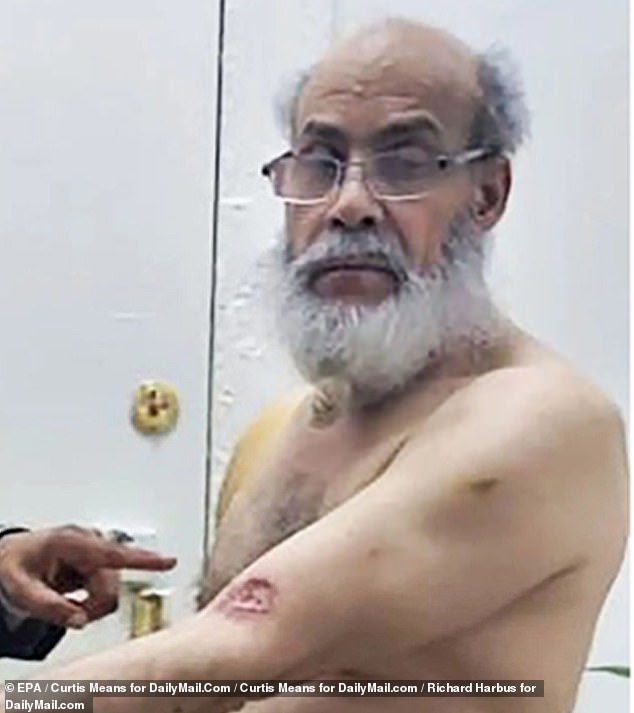
José Alba shows the injuries he suffered during the brutal attack in his winery

Alba (arm in the photo) shows photographers her wounds with her friend Francisco Marte
Other videos obtained by DailyMail.com show Simon, who was on probation for attacking a police officer when he died, marching behind the bodega counter to confront Alba.
‘Dad, I don’t want problems, dad,’ Alba told him calmly.
Simon burst into the Blue Moon store minutes after his girlfriend tried to buy a bag of chips for her daughter, but her e-benefits card had been declined.

