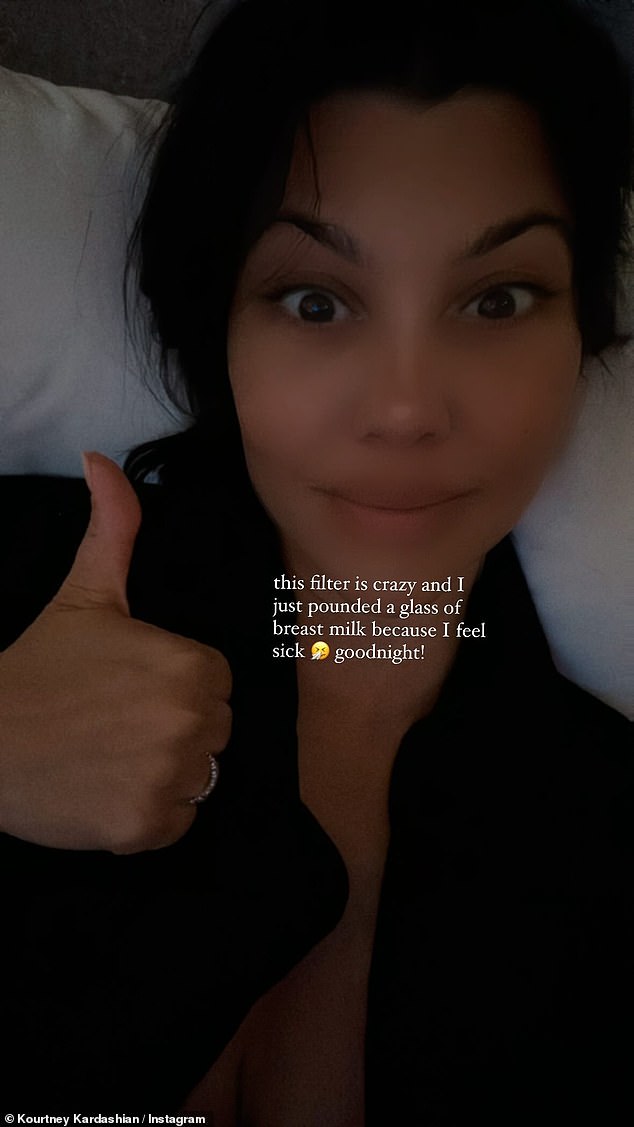
Kourtney Kardashian has caused confusion among her fans by admitting that she drank an entire glass of her own breast milk.
No stranger to a peculiar health trend, the Keeping Up With The Kardashians star confessed that she turned to drinking after feeling sick for several hours.
Kourtney had been breastfeeding her five-month-old son Rocky Thirteen and has talked about maximizing her milk supply by drinking amaranth, barley, congee, millet, oats and rice, as well as bone broth, bone broth stews, kichari and rice soup.
Posting a selfie of her lying in bed, Kourtney captioned her post: “This filter is crazy and I just drank a glass of breast milk because I feel sick.” good night.’
Kourtney previously shared a photo of herself pumping breast milk in sexy black lingerie, after welcoming her son with husband Travis Barker last year.
Kourtney Kardashian has caused confusion among her fans by admitting that she drank an entire glass of her own breast milk.

The star previously shared a photo of herself pumping breast milk in a low-cut black dress, after welcoming her son Rocky with husband Travis Barker last year.
The post sparked a mixed reaction from viewers, with some wondering why she would share a photo of herself ‘expressing breast milk’.
The star also gave insight into her post-baby health regime, returning to the gym for the first time seven weeks after the birth of her son.
Kourtney wrote in the video that she was “doing the most important job in the world…being a mom: keeping my body alive, breastfeeding on demand means keeping calories up with lots of nutritious foods.”
He noted that he was “taking the time to slowly return to some uphill walks on the treadmill when I can sneak them in, along with my walks outside in the stroller.”
Kourtney is far from the first big name to try drinking breast milk, with Brody Jenner admitting last year that he uses it in his morning coffee.
The Hills alum shared a clip of himself making a cup of coffee for himself and his fiancee Tia Blanco, using the unconventional ingredient as cream.
In a new YouTube video, Brody discovered that he had run out of almond milk while making lattes, but discovered that breast milk was an amazing alternative.
Serena Williams also revealed in December that she donated the excess breast milk she had to mothers and babies in need.

The 44-year-old and her husband Travis Barker announced in early November that she had given birth to their son Rocky Thirteen.

She is one of many stars who have followed this strange health trend, and Ferne McCann also admits she has tried her own breast milk.

Brody Jenner also admitted last year that he used some of his fiancee’s breast milk in his morning coffee after running out of almond milk.
In October, British reality star Ferne McCann also surprised Steph’s Packed Lunch viewers when she appeared on the show to talk about her six-month-old daughter, Finty, and revealed that she had also tried her own breast milk.
Ferne, who is also a doting mum to her six-year-old daughter Sunday, who she shares with ex Arthur Collins, said her breast milk was like “liquid gold”.
He also revealed that the nurses told him to use it as a first aid remedy. “Finty, when she was born, had a little bit of tears in her eyes, and the midwives in the community were like, ‘Give her some breast milk,'” the model explained.
“If Lorri had a blister, I would suggest squeezing a little on the blister, it would heal in a few moments.”
Bodybuilders have been known to try drinking breast milk in a bid to build their biceps, although experts have stated that drinking breast milk is only safe if it comes from a partner.
This is because breast milk is a body fluid, which means that adults who drink it could be at risk of contracting infectious diseases such as cytomegalovirus, hepatitis B and C, human immunodeficiency virus or syphilis.
The US Food and Drug Administration (FDA) also previously warned about the dangers of giving a baby breast milk from a source other than the child’s mother.
“Risks to the baby include exposure to infectious diseases, including HIV, to chemical contaminants, such as some illegal drugs, and to a limited number of prescription drugs that may be in human milk,” according to the FDA website.
Additionally, milk can become contaminated and unsafe to drink if not handled and stored properly. Adults are also susceptible to some of the same risks if they drink another person’s breast milk.